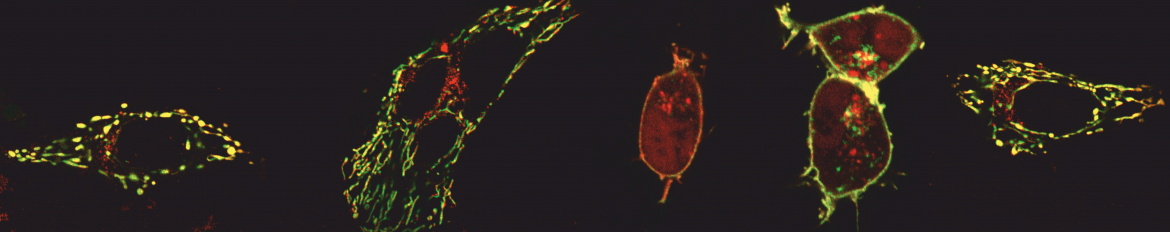
Calcium ions are central and ubiquitous intracellular signalling entities. Changes in intracellular calcium levels control fundamental biological events, from initiating the mitotic birth of a new cell to marking its end during apoptosis. Intracellular calcium signals in mammalian cells are initiated primarily through opening of calcium channels found either in the plasma membrane (PM) or in the membrane of intracellular calcium stores, i.e., the endoplasmic reticulum (ER). Our lab studies an exceptional cellular process called store-operated calcium entry, whereby the opening of calcium channels in the ER, which lead to depletion of this intracellular calcium store, triggers the opening of calcium channels in the PM.
The classical store operated channel is the Calcium Release-Activated Calcium (CRAC) channel. This channel is formed by STIM and Orai proteins and its activity controls key cellular processes such as gene expression, cell differentiation, secretion, and Ca2+ homeostasis. Calcium signals generated by CRAC channels are particularly important for executing the immune functions of T and B lymphocytes and loss of function mutations in the STIM1 or Orai1 genes leads to a severe form of immunodeficiency in humans.
[pb_slideshow group=”1″]We are currently pursuing two main research directions. In the first we investigate how coupling between STIM and Orai proteins controls channel opening and how additional interactions with SARAF regulate this process. In the second we develop novel synthetic photo-switchable compounds that control CRAC channel activity by light. We currently explore the usage of such compounds as research tools and as optically controlled drugs.
For these studies we combine genetic, protein and chemical engineering with patch-clamp electrophysiology and high resolution imaging using total internal reflection fluorescence (TIRF), confocal, and single-molecule microscopy.
While mitochondria are mostly recognized for their metabolic functions, such as ATP and lipid synthesis, they also constitute an important intracellular signaling hub. We are studying the molecular mechanisms of two interrelated aspects of mitochondrial biology: mitochondrial calcium signaling and mitochondrial heterogeneity.
Mitochondrial Ca2+ signalling
The uptake and release of Ca2+ by mitochondrial regulates energy production, shapes cytosolic Ca2+ signals and determines cell death or survival. The molecules that directly control the passage of calcium across the mitochondrial inner membrane remained a mystery for close to 50 years. In recent major breakthrough discoveries, we identified the NCLX gene and others have identified the MCU gene, key components mediating mitochondrial calcium release and uptake, respectively. Yet, the complete molecular representation of mitochondrial cation transport mechanisms remains incomplete. We seek to fill this knowledge gap and to understand how the combined workings of multiple mitochondrial cation transporters affect mitochondrial and cellular functions.
Mitochondrial contact sites
In most cell types a subpopulations of mitochondria localize to different cellular compartments like the PM and ER membranes. This cellular organization enables mitochondria to play central roles in shaping intracellular Ca2+ signaling, lipid synthesis, cellular metabolism, autophagy and in apoptosis progression. We are interested in elucidating the molecular mechanisms responsible for the close juxta-positioning between mitochondria and membranes of the ER or PM.
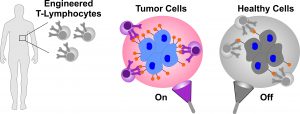
[pb_slideshow group=”2″]Cell-based immunotherapies are emerging as promising treatments for cancer. Leading immunotherapy approaches involve genetic engineering and reprogramming of the patients’ own T cells in order to stimulate and direct them to combat cancer cells. These approaches, however, are currently limited because tumor-specific antigens are not easily identified and because targeting of non-cancer cells together with the hyper-immune reaction of the engineered T cells often leads to various adverse effects including death. Thus, a pressing challenge of T cell-mediated immunotherapies is to achieve selective targeting of cancer cells together with a tunable and reversible control of the immune reaction in order to reduce both off-target and adverse effects. To address these issues, we are engineering an on-off safety switch that utilizes specific kinds of light to allow for T cell activation to occur only where, when and at the precise intensity needed.